Abstract
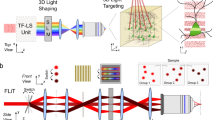
Light-sensitive 'caged' molecules provide a means of rapidly and noninvasively manipulating biochemical signals with submicron spatial resolution. Here we describe a new optical system for rapid uncaging in arbitrary patterns to emulate complex neural activity. This system uses TeO2 acousto-optical deflectors to steer an ultraviolet beam rapidly and can uncage at over 20,000 locations per second. The uncaging beam is projected into the focal plane of a two-photon microscope, allowing us to combine patterned uncaging with imaging and electrophysiology. By photolyzing caged neurotransmitter in brain slices we can generate precise, complex activity patterns for dendritic integration. The method can also be used to activate many presynaptic neurons at once. Patterned uncaging opens new vistas in the study of signal integration and plasticity in neuronal circuits and other biological systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
28 November 2005
In the version of this article initially published online, Supplementary Videos 1 and 2 were in AVI format. These videos have been replaced with new versions in MOV format.
References
Polsky, A., Mel, B.W. & Schiller, J. Computational subunits in thin dendrites of pyramidal cells. Nat. Neurosci. 7, 621–627 (2004).
London, M. & Segev, I. Synaptic scaling in vitro and in vivo. Nat. Neurosci. 4, 853–855 (2001).
Adams, S.R. & Tsien, R.Y. Controlling cell chemistry with caged compounds. Annu. Rev. Physiol. 55, 755–784 (1993).
Callaway, E.M. & Yuste, R. Stimulating neurons with light. Curr. Opin. Neurobiol. 12, 587–592 (2002).
Heidelberger, R., Heinemann, C., Neher, E. & Matthews, G. Calcium dependence of the rate of exocytosis in a synaptic terminal. Nature 371, 513–515 (1994).
DelPrincipe, F., Egger, M. & Niggli, E. Calcium signalling in cardiac muscle: refractoriness revealed by coherent activation. Nat. Cell Biol. 1, 323–329 (1999).
Jones, K.T. & Nixon, V.L. Sperm-induced Ca2+ oscillations in mouse oocytes and eggs can be mimicked by photolysis of caged inositol 1,4,5-trisphosphate: evidence to support a continuous low level production of inositol 1,4,5-trisphosphate during mammalian fertilization. Dev. Biol. 225, 1–12 (2000).
Echevarría, W., Leite, M.F., Guerra, M.T., Zipfel, W.R. & Nathanson, M.H. Regulation of calcium signals in the nucleus by a nucleoplasmic reticulum. Nat. Cell Biol. 5, 440–446 (2003).
Goard, M. et al. Light-mediated inhibition of protein synthesis. Chem. Biol. 12, 685–693 (2005).
Bullen, A., Patel, S.S. & Saggau, P. High-speed, random-access fluorescence microscopy: I. High-resolution optical recording with voltage-sensitive dyes and ion indicators. Biophys. J. 73, 477–491 (1997).
Saggau, P., Bullen, A. & Patel, S.S. Acousto-optic random-access laser scanning microscopy: fundamentals and applications to optical recording of neuronal activity. Cell Mol. Biol. 44, 827–846 (1998).
Shepherd, G.M., Pologruto, T.A. & Svoboda, K. Circuit analysis of experience-dependent plasticity in the developing rat barrel cortex. Neuron 38, 277–289 (2003).
Matsuzaki, M., Honkura, N., Ellis-Davies, G.C.R. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004).
Denk, W., Strickler, J.H. & Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990).
Tsai, P.S. et al. Principles, design and construction of a two-photon laser-scanning microscope for in vitro and in vivo studies. In In vivo optical imaging of brain function (ed. Frostig, R.D.) 113–171 (CRC Press, Boca Raton, Florida, USA, 2002).
Iyer, V., Losavio, B.E. & Saggau, P. Compensation of spatial and temporal dispersion for acousto-optic multiphoton laser-scanning microscopy. J. Biomed. Opt. 8, 460–471 (2003).
Bliton, A.C. & Lechleiter, J.D. Optical considerations at ultraviolet wavelengths in confocal microscopy. In Handbook of biological confocal microscopy (ed. Pawley, J.B.) 431–444 (2nd edn.Plenum Press, New York, 1995).
Brown, E. & Webb, W. Two-photon activation of caged calcium with submicron, submillisecond resolution. in Caged Compounds. Methods of Enzymology. (ed. Marriott, G.) 291, 356–380 (Academic Press, San Diego, 1998).
Papageorgiou, G. & Corrie, J.E.T. Effects of aromatic substituents on the photocleavage of 1-acyl-7-nitroindolines. Tetrahedron 56, 8197–8205 (2000).
Canepari, M., Nelson, L., Papageorgiou, G., Corrie, J.E. & Ogden, D. Photochemical and pharmacological evaluation of 7-nitroindolinyl-and 4-methoxy-7-nitroindolinyl-amino acids as novel, fast caged neurotransmitters. J. Neurosci. Methods 112, 29–42 (2001).
Matsuzaki, M. et al. Dendritic spine geometry is critical for AMPA receptor expression in hippocampal CA1 pyramidal neurons. Nat. Neurosci. 4, 1086–1092 (2001).
Smith, M.A., Ellis-Davies, G.C. & Magee, J.C. Mechanism of the distance-dependent scaling of Schaffer collateral synapses in rat CA1 pyramidal neurons. J. Physiol. (Lond.) 548, 245–258 (2003).
O'Connor, D.H., Wittenberg, G.M. & Wang, S.S-H. Graded bidirectional synaptic plasticity is composed of switch-like unitary events. Proc. Natl. Acad. Sci. USA 102, 9679–9684 (2005).
Schiller, J. & Schiller, Y. NMDA receptor-mediated dendritic spikes and coincident signal amplification. Curr. Opin. Neurobiol. 11, 343–348 (2001).
Callaway, E.M. & Katz, L.C. Photostimulation using caged glutamate reveals functional circuitry in living brain slices. Proc. Natl. Acad. Sci. USA 90, 7661–7665 (1993).
Barbour, B. Synaptic currents evoked in Purkinje cells by stimulating individual granule cells. Neuron 11, 759–769 (1993).
Chadderton, P., Margrie, T.W. & Häusser, M. Integration of quanta in cerebellar granule cells during sensory processing. Nature 428, 856–860 (2004).
Isope, P., Franconville, R., Barbour, B. & Ascher, P. Repetitive firing of rat cerebellar parallel fibres after a single stimulation. J. Physiol. (Lond.) 554, 829–839 (2004).
Kandler, K., Katz, L.C. & Kauer, J.A. Focal photolysis of caged glutamate produces long-term depression of hippocampal glutamate receptors. Nat. Neurosci. 1, 119–123 (1998).
Dodt, H., Eder, M., Frick, A. & Zieglgansberger, W. Precisely localized LTD in the neocortex revealed by infrared-guided laser stimulation. Science 286, 110–113 (1999).
Eder, M., Zieglgansberger, W. & Dodt, H.U. Neocortical long-term potentiation and long-term depression: site of expression investigated by infrared-guided laser stimulation. J. Neurosci. 22, 7558–7568 (2002).
Abbott, L.F. & Nelson, S.B. Synaptic plasticity: taming the beast. Nat. Neurosci. 3, 1178–1183 (2000).
Froemke, R.C., Poo, M-m. & Dan, Y. Spike-timing-dependent synaptic plasticity depends on dendritic location. Nature 434, 221–225 (2005).
Stuart, G.J. & Häusser, M. Dendritic coincidence detection of EPSPs and action potentials. Nat. Neurosci. 4, 63–71 (2001).
Koh, I.Y., Lindquist, W.B., Zito, K., Nimchinsky, E.A. & Svoboda, K. An image analysis algorithm for dendritic spines. Neural Comput. 14, 1283–1310 (2002).
Hyland, B.I., Reynolds, J.N., Hay, J., Perk, C.G. & Miller, R. Firing modes of midbrain dopamine cells in the freely moving rat. Neuroscience 114, 475–492 (2002).
Brivanlou, I.H., Dantzker, J.L., Stevens, C.F. & Callaway, E.M. Topographic specificity of functional connections from hippocampal CA3 to CA1. Proc. Natl. Acad. Sci. USA 101, 2560–2565 (2004).
Horch, K.W. & Dhillon, G.S. (eds.). Neuroprosthetics: theory and practice (World Scientific, New Jersey, 2004).
Acknowledgements
This work was supported by grants from the Princeton Center for Photonics and Optoelectronic Materials, the US National Institutes of Health, the National Science Foundation (NSF), and the W.M. Keck Foundation to S.S.-H.W., a Lewis Thomas fellowship to S.S., an NSF Graduate Research Fellowship to D.H.O. and a Burroughs-Wellcome Interfaces of Science fellowship to D.V.S. We thank J. Soos from Brimrose Corp. for designing the AOD device and K. Visscher, M. McDonald, J. Puchalla, G. Wittenberg, G. Major and T. Adelman for advice and discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Axial and lateral resolution depends on depth in brain slices. (PDF 82 kb)
Supplementary Fig. 2
Patterned uncaging and fluorescence measurement of intracellular calcium signals. (PDF 112 kb)
Supplementary Video 1
Current summation in a pyramidal neuron. In the first video segment, uncaging locations are shown in blue on a two-photon image of a dye-filled CA1 pyramidal neuron. Voltage-clamp current responses to a complex uncaging pattern are highly reproducible when the stimulation pattern is repeated (first three current traces, shown in color). In contrast, the current response is different when a new stimulation pattern with the same mean rate is delivered across the same dendritic locations (fourth current trace, shown in black). In the second video segment, a freely-spiking pyramidal neuron shows highly reproducible voltage responses to identical stimuli (first three voltage traces, shown in color). A different stimulus with the same mean rate gives a different voltage response (fourth voltage trace, shown in black). (MOV 1779 kb)
Supplementary Video 2
IP3 uncaging in a Purkinje neuron. Ca2+ release responses are shown after patterned release of caged IP3 at six locations in a rat cerebellar Purkinje neuron. The Purkinje cell was filled through a patch-pipette with 100 μM double-caged IP3 and 300 μM of the Ca2+-sensitive dye fluo-5F, and imaged with a two-photon microscope at a rate of 32 ms per frame. The first segment of the video shows responses to uncaging at 1-second intervals. The second segment shows responses to uncaging at the same locations at 320-millisecond intervals. (MOV 1735 kb)
Supplementary Data 1
Axial resolution in brain slices. (PDF 24 kb)
Supplementary Data 2
Calcium release in response to focal uncaging. (PDF 25 kb)
Supplementary Note
Optical system description and plan. (PDF 269 kb)
Rights and permissions
About this article
Cite this article
Shoham, S., O'Connor, D., Sarkisov, D. et al. Rapid neurotransmitter uncaging in spatially defined patterns. Nat Methods 2, 837–843 (2005). https://doi.org/10.1038/nmeth793
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth793
This article is cited by
-
Rationally designed azobenzene photoswitches for efficient two-photon neuronal excitation
Nature Communications (2019)
-
Optogenetic stimulation of complex spatio-temporal activity patterns by acousto-optic light steering probes cerebellar granular layer integrative properties
Scientific Reports (2018)
-
A distinct entorhinal cortex to hippocampal CA1 direct circuit for olfactory associative learning
Nature Neuroscience (2017)
-
An implantable microelectrode array for simultaneous L-glutamate and electrophysiological recordings in vivo
Microsystems & Nanoengineering (2015)
-
Holographic optogenetic stimulation of patterned neuronal activity for vision restoration
Nature Communications (2013)