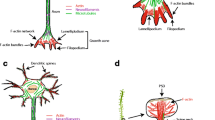
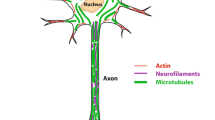
Abstract
The proline-directed serine threonine kinase, Cdk5, is an unusual molecule that belongs to the well-known large family of proteins, cyclin-dependent kinases (Cdks). While it has significant homology with the mammalian Cdk2 and yeast cdc2, unlike the other Cdks, it has little role to play in cell cycle regulation and is activated by non-cyclin proteins, p35 and p39. It phosphorylates a spectrum of proteins, most of them associated with cell morphology and motility. A majority of known substrates of Cdk5 are cytoskeletal elements, signalling molecules or regulatory proteins. It also appears to be an important player in cell–cell communication. Highly conserved, Cdk5 is most abundant in the nervous system and is of special interest to neuroscientists as it appears to be indispensable for normal neural development and function. In normal cells, transcription and activity of Cdk5 is tightly regulated. Present essentially in post-mitotic neurons, its normal activity is obligatory for migration and differentiation of neurons in developing brain. Deregulation of Cdk5 has been implicated in Alzheimer’s disease, amyotrophic lateral sclerosis, Parkinson’s disease, Huntington’s disease and acute neuronal injury. Regulators of Cdk5 activity are considered as potential therapeutic molecules for degenerative diseases. This review focuses on the role of Cdk5 in neural cells as regulator of cytoskeletal elements, axonal guidance, membrane transport, synaptogenesis and cell survival in normal and pathological conditions.

Similar content being viewed by others
References
Alexander K, Yang H-S, Hinds PW (2004) Cellular senescence requires CDK5 repression of Rac1 activity. Mol Cell Biol 24:2808–2819
Altucci L, Addeo R, Cicatiello L, Germano D, Pacilio C, Battista T, Cancemi M, Petrizzi VB, Bresciani F, Weisz A (1997) Estrogen induces early and timed activation of cyclin-dependent kinases 4, 5, and 6 and increases cyclin messenger ribonucleic acid expression in rat uterus. Endocrinology 138(3):978–984
Augustinack JC, Sanders JL, Tsai LH, Hyman BT (2002) Colocalization and fluorescence resonance energy transfer between cdk5 and AT8 suggests a close association in pre-neuro.brillary tangles and neuro.brillary tangles. J Neuropathol Exp Neurol 61:557–564
Barclay JW, Aldea M, Craig TJ, Morgan A, Burgoyne RD (2004) Regulation of the fusion pore conductance during exocytosis by cyclin-dependent kinase 5. J Biol Chem 279(40):41495–41503
Bhaskar K, Mohammed MS, Sharma VM, Pavan KSA, Ramamohan Y, Pant HC, Raju TR, Taranath SK (2003) Co-purification and localization of munc-18–1 (P67) and Cdk5 with neuronal cytoskeletal proteins. Neurochem Int 44(1):35–44
Bibb JA, Chen J, Taylor JR et al (2001) Effects of chronic exposure to cocaine are regulated by the neuronal protein Cdk5. Nature 410:376–380
Bibb JA (2003a) Role of Cdk5 in neuronal signaling, plasticity, and drug abuse. Neurosignals 12:191–199
Bibb JA, Nguyen C (2003b) Cdk5 and the mystery of synaptic vesicle endocytosis. J Cell Biol 163(4):697–699
Borghi R, Giliberto L, Assini A, Delacourte A, Perry G, Smith MA, Strocchi P, Zaccheo D, Tabaton M (2002) Increase of cdk5 is related to neuro.brillary pathology in progressive supranuclear palsy. Neurology 58:589–592
Brion JP, Couck AM (1995) Cortical and brainstem-type Lewy bodies are immunoreactive for the cyclin-dependent kinase 5. Am J Pathol 147:1465–1476
Brown NR, Noble ME, Endicott JA, Johnson LN (1999) The structural basis for specificity of substrate and recruitment peptides for cyclin-dependent kinases. Nat Cell Biol 1:438
Bu B, Li J, Davies P, Vincent I (2002) Deregulation of cdk5, hyperphosphorylation, and cytoskeletal pathology in the Niemann-pick type C murine model. J Neurosci 22:6515–6525
Buzko O, Shokat KM (2002) A kinase sequence database: sequence alignments and family assignment. Bioinformatics 18:1274; Nature 1995 374:131
Camins A, Verdaguer E, Folch J, Canudas AM, Pallas M (2006) The role of CDK5/P25 formation/inhibition in neurodegeneration. Drug News Perspect 19(8):453–460
Chae T, Kwon YT, Bronson R, Dikkes P, Li E, Tsai LH (1997) Mice lacking p35, a neuronal specific activator of Cdk5, display cortical lamination defects, seizures, and adult lethality. Neuron 18(1):29–42
Chen YZ, Kelz MB. Steffen C, Ang ES, Zeng L, Nestler EJ (2000) Induction of cyclin-dependent kinase 5 in the hippocampus by chronic electroconvulsive seizures: role of dFosB. J Neurosci 20:8965–8971
Chen F, Studzinski GP (2001) Expression of the neuronal cyclin-dependent kinase 5 activator p35Nck5a in human monocytic cells is associated with differentiation. Blood 97(12):3763–3767
Cheng K, Ip NY (2003) Cdk5: a new player at synapses. Neurosignals 12:180–190
Cheng K, Li Z, Fu W-Y, Wang JH, Fu AKY, Ip NY (2002) Pctaire1 interacts with p35, is a novel substrate for Cdk5/p35. J Biol Chem 277 (35):31988–31993
Cheung ZH, Chin WH, Chen Y, Ng YP, Ip NY (2007a) Cdk5 Is involved in BDNF-stimulated dendritic growth in hippocampal neurons. PLoS Biol 5(4):e63
Cheung ZH, Ip NY (2007b) The roles of Cdk5 in dendrite and synapse development. Biotechnol J 2(8):949–957
Cicero S, Herrup K (2005) Cdk5 is essential for neuronal cell cycle arrest and differentiation. J Neurosci 25(42):9658–9668
Contractor et al (2002) Trans-synaptic eph receptor-ephrin signaling in hippocampal mossy fiber LTP. Science’s STKE 7:1864
Cruz J, Tseng H, Goldman J, Shih H, Tsai L (2003) Aberrant Cdk5 activation by p25 triggers pathological events leading to neurodegeneration and neurofibrillary tangles. Neuron 40(3):471–483
Cruz J, Tsai L-H (2004a) A Jekyll and Hyde kinase: roles for Cdk5 in brain development and disease. Curr Opin Neurobiol 14(3):390–394
Cruz J, Tsai L-H (2004b) Cdk5 deregulation in the pathogenesis of Alzheimer’s disease. Trends Mol Med 10(9):452–458
Damu T, Jeffery Y, Ki-Young L, Masayuki M, Hideki M, Kazuhito T, Osamu H, Jerry HW (1996) An isoform of the neuronal cyclin-dependent kinase 5 (Cdk5) activator. J Biol Chem 270(45):26897–26903
Delalle I, Bhide PG, Caviness VS Jr, Tsai L-H (1997) Temporal and spatial patterns of expression of p35, a regulating subunit of Cdk5 in NS of mouse. J Neurocytol 26(5):283–296
Dhavan R, Tsai L-H (2001) Decade of Cdk5. Nat Rev Mol Cell Biol 2(10):749–759
Fahn S (2003) Description of Parkinson’s disease as a clinical syndrome. Ann NY Acad Sci 991:1–14
Fischer A, Sananbenesi F, Schrick C, Spiess J, Radulovic J (1997) Cyclin-dependent kinase 5 is required for associative learning. J Neurosci 22(9):3700–3707
Fischer A, Sananbenesi F, Pang PT, Lu B, Tsai L-H (2005) Opposing roles of transient and prolonged expression of p25 in synaptic plasticity and hippocampus-dependent memory. Neuron 48:825–838
Flaherty DB, Soria JP, Tomasiewicz HG, Wood JG (2000) Phosphorylation of human tau protein by microtubule-associated kinases: GSK3β and cdk5 are key participants. J Neurosci Res 62(3):463–472
Fletcher AI, Shuang R, Giovannucci DR, Zhang L, Bittner MA et al (1999) Regulation of exocytosis by cyclin-dependent kinase 5 via phosphorylation of Munc18. J Biol Chem 274:4027–4035
Fu AK, Fu WY, Cheung J, Tsim KW, Ip FC et al (2001) Cdk5 is involved in neuregulin-induced AChR expression at the neuromuscular junction. Nat Neurosci 4:374–381
Fu AK, Fu WY, Ng AK, Chien WW, Ng YP et al (2004) Cyclin-dependent kinase 5 phosphorylates signal transducer and activator of transcription 3 and regulates its transcriptional activity. Proc Natl Acad Sci USA 101:6728–6733
Fu WY, Fu AK, Lok KC, Ip FC, Ip NY (2002) Induction of Cdk5 activity in rat skeletal muscle after nerve injury. Neuroreport 13:243–247
Fu W-Y, Chen Y, Sahin M, Zhao X-S, Shi L, Bikoff JB, Lai K-O, Yung W-H, Fu AK, Greenberg ME, Ip NY (2006) Cdk5 regulates EphA4-mediated dendritic spine retraction through an ephexin1-dependent mechanism. Nat Neurosci 10(1):67–76
Gao CY, Zakeri Z, Zhu Y, He H, Zelenka PS (1997) Expression of Cdk5, p35, and Cdk5-associated kinase activity in the developing rat lens. Dev Genet 20:267–275
Gao CY, Negash S, Wang HS, Ledee D, Guo H, Russell P, Zelenka P (2001) Cdk5 mediates changes in morphology and promotes apoptosis of astrocytoma cells in response to heat shock. J Cell Sci 114:1145–1153
Gao C, Negash S, Guo HT, Ledee D, Wang H-S, Zelenka P (2002a) Cdk5 regulates cell adhesion and migration in corneal epithelial cells. Mol Cancer Res 1:12–24
Gao C, Negash S, Guo HT, Ledee D, Wang H-S, Zelenka P (2002b) CDK5 regulates cell adhesion and migration in corneal epithelial cells. Mol Cancer Res 1:12–24
Gao CY, Stepp MA, Fariss R, Zelenka P (2004) Cdk5 regulates activation and localization of Src during corneal epithelial wound closure. J Cell Sci 117:4089–4098
Gilmore EC, Ohshima T, Goffinet AM, Kulkarni AB, Herrup K (1998) Cyclin-dependent kinase 5-deficient mice demonstrate novel developmental arrest in cerebral Cortex. J Neurosci 18(16):6370–6377
Gong X, Tang X, Wiedmann M, Wang X, Peng J et al (2003) Cdk5-mediated inhibition of the protective effects of transcription factor MEF2 in neurotoxicity-induced apoptosis. Neuron 38:33–46
Grant P, Sharma P, Pant HC (2001a) Cyclin-dependent protein kinase 5 (Cdk5) and the regulation of neurofilament metabolism. Eur J Biochem 268:1534–1546
Green SL, Kulp KS, Vulliet R (1997) Cyclin-dependent protein kinase 5 activity increases in rat brain following ischemia. Neurochem Int 31(4):617–623
Green SL, Vulliet PR, Pinter MJ, Cork LC (1998) Alterations in cyclin-dependent protein kinase 5 (CDK5) protein levels, activity and immunocytochemistry in canine motor neuron disease. J Neuropathol Exp Neurol 57:1070–1077
Grunwald IC, Korte M, Adelmann G, Plueck A, Kullander K, Adams R, Frotscher M, Bonhoeffer T, Klein R (2004) Hippocampal plasticity requires postsynaptic ephrinBs. Nat Neurosci 7:33–40
Hallows JL, Chen K, DePinho RA, Vincent I (2003) Decreased cyclin-dependent kinase 5 (Cdk5) activity is accompanied by redistribution of Cdk5 and cytoskeletal proteins and increased cytoskeletal protein phosphorylation in P35 null mice. J Neurosci 23(33):10633–10644
Hamdane M, Sambo A-V, Delobel P, Bégard S, Violleau A, Delacourte A, Bertrand P, Benavides J, Buée L (2003) Mitotic-like tau phosphorylation By P25-Cdk5 kinase complex. J Biol Chem 2780(36):34026–34034
Hamdane M, Bretteville A, Sambo A-V, Schindowski K, Bégard S, Delacourte A, Bertrand P, Buée L (2005) P25/Cdk5 mediated retinoblastoma phosphorylation is an early event in neuronal cell death. J Cell Sci 118:1291–1298
Hellmich MR, Pant HC, Wada E, Battey JF (1992) Neuronal cdc2-like kinase: a cdc2-related protein kinase with predominantly neuronal expression. Proc Natl Acad Sci USA 89:10867–10871
Henkemeyer M, Itkis OS, Ngo M, Hickmott PW, Ethell IM (2003) Multiple EphB receptor tyrosine kinases shape dendritic spines in the hippocampus. J Cell Biol 163:1313–1326
Hirasawa M, Ohshima T, Takahashi S, Longenecker G, Honjo Y, Veeranna, Pant HC, Mikoshiba K, Brady RO, Kulkarni AB (2004) Perinatal abrogation of Cdk5 expression in brain results in neuronal migration defects. PNAS 101(16):6249–6254
Honjyo Y, Kawamoto Y, Nakamura S, Nakano S, Akiguchi I (2001) P39 immunoreactivity in glial cytoplasmic inclusions in brains with multiple system atrophy. Acta Neuropathol (Berlin) 101:190–194
Iijima K, Ando K, Takeda S, Satoh Y, Seki T, Itohara S, Greengard P, Kirino Y, Nairn AC, Suzuki T (2000) Neuron-specific phosphorylation of Alzheimer’s beta-amyloid precursor protein by cyclin-dependent kinase 5. J Neurochem 75:1085–1091
Ishiguro K, Takamatsu M, Tomizawa K, Omori A, Takahashi M, Arioka M, Uchida T, Imahori K (1992) Tau protein kinase I converts normal tau protein into A68-like component of paired helical filaments. J Biol Chem 267:10897–10901
Kamei H, Saito T, Ozawa M, Fujita Y, Asada A, Bibb J, Saido TC, Sorimachi H, Hisanaga S-i (2007) Suppression of calpain-dependent cleavage of the CDK5 activator p35 to p25 by site-specific phosphorylation. J Biol Chem 282(3):1687–1694
Kazuya S, Alka A-M, Li W, Wei S, Yutaka M, Hemant KP (2000) Interaction of neuronal Cdc2-like protein kinase with microtubule-associated protein tau. J Biol Chem 275(22):16673–16680
Kerokoski P, Suuronen T, Salminen A, Soininen H, Pirttila T (2001) The levels of cdk5 and p35 proteins and tau phosphorylation are reduced during neuronal apoptosis. Biochem Biophys Res Commun 280:998–1002; 101:190–194
Kerokoski P, Suuronen T, Salminen A, Soininen H, Pirttila T (2002) Cleavage of the cyclin-dependent kinase 5 activator p35 to p25 does not induce tau hyperphosphorylation. Biochem Biophys Res Commun 298:693–698
Ko J, Humbert S, Bronson RT, Takahashi S, Kulkarni AB, Li E, Tsai LH (2001) P35 and P39 are essential for cyclin-dependent kinase 5 function during neurodevelopment. J Neurosci 21(17):6758–6771
Kobayashi S, Ishiguro K, Omori A, Takamatsu M, Arioka M, Imahori K, Uchida T (1993) A cdc2-related kinase PSSALRE/cdk5 is homologous with the 30 kDa subunit of tau protein kinase II, a proline-directed protein kinase associated with microtubule. FEBS Lett 335:171–175
Krieger C, Hu JH, Pelech S (2003) Aberrant protein kinases and phosphoproteins in amyotrophic lateral sclerosis. Trends Pharmacol Sci 24(10):535–541
Lazaro JB, Kitzmann M, Poul MA, Vandromme M, Lamb NJ, Fernandez A (1997) Cyclin dependent kinase 5, cdk5, is a positive regulator of myogenesis in mouse C2 cells. J Cell Sci 110(10):1251–1260
Lee KY, Helbing CC, Choi KS, Johnston RN, Wang JH (1997) Neuronal Cdc2-like kinase (Nclk) binds and phosphorylates the retinoblastoma protein. J Biol Chem 272(9):5622–5626
Lee M-s, Kwon Young T, Li Mingwei, Peng Junmin, Friedlander Robert M, Tsai Li-Huei (2000) Neurotoxicity induced cleavage of P35 to P25 by calpain. Nature 405:360–364
Lee MS, Nikolic M, Baptista CA, Lai E, Tsai LH, Massague J (1996) The brain-specific activator p35 allows Cdk5 to escape inhibition by p27Kip1 in neurons. Proc Natl Acad Sci USA 93:3259
Lee M-S, Tsai L-H (2001) Cdk5 at the Junction. Nat Neurosci 4:340–342
Lee M-S, Tsai L-H (2003) Cdk5: one of the links between senile plaques and neurofibrillary tangles? J Alzheimer’s Dis 5(2):127–137
Lew J, Beaudette K, Litwin CM, Wang JH (1992) Purification and characterization of a novel proline-directed protein kinase from bovine brain. J Biol Chem 267:13383–13390
Lew J, Huang QQ, Qi Z, Winkfein RJ, Aebersold R, Hunt T, Wang JH (1994) A brain-specific activator of cyclin-dependent kinase 5. Nature 371:423–426
Li BS, Zhang L, Gu J, Amin ND, Pant H (2000) Integrin α1β1 mediated activation of Cdk5 activity is involved in neurite outgrowth and human neurofilament protein H Lys Ser Pro tail domain phosphorylation. J Neurosci 20(16):6055–6062
Li B-S, Sun M-K, Zhang L, Takahashi S, Ma W, Vinade L, Kulkarni AB, Brady RO, Pant HC (2001) Regulation of NMDA receptors by cyclin-dependent kinase-5. PNAS 98(22):12742–12747
Li B-S, Ma W, Jaffe H, Zheng Y, Takahashi S, Zhang L, Kulkarni AB, Pant HC (2003) Cyclin-dependent kinase-5 is involved in neuregulin-dependent activation of phosphatidylinositol 3-kinase and Akt activity mediating neuronal survival. J Biol Chem 278(37):35702–35709
Lilja L, Yang SN, Webb DL, Juntti-Berggren L, Berggren PO et al (2001) Cyclin-dependent kinase 5 promotes insulin exocytosis. J Biol Chem 276:34199–34205
Lilja L, Johansson JU, Gromada J, Mandic SA, Fried G et al (2004) Cyclin-dependent kinase 5 associated with p39 promotes Munc18–1 phosphorylation and Ca(2þ)-dependent exocytosis. J Biol Chem 279:29534–29541
Lin H, Juang JL, Wang PS (2004) Involvement of Cdk5/p25 in digoxin-triggered prostate cancer cell apoptosis. J Biol Chem 279(28):29302–29307
Maccioni RB, Otth C, Concha II, Munoz JP (2001) The protein kinase Cdk5: structural aspects in neurogenesis and involvement in Alzheimer’s pathology. Eur J Biochem 268(10):1518–1527
Manning G, Whyte DB, Martinez R, Hunter T, Sudarsanam S (2002) The protein kinase complement of the human genome. Science 298:1912
Mapelli M, Musacchio A (2003) The structural perspective on Cdk5. Neurosignals 12:164–172
Meyerson M, Enders GH, Wu CL, Su LK, Gorka C, Nelson C, Harlow E, Tsai LH (1992) A family of human cdc2-related protein kinases. EMBO J 11:2909–2917
Morgan D (1995) Principals of Cdk regulation. Nature 374:131–134
Musa FR, Tokuda M, Kuwata Y, Ogawa T, Tomizawa K et al (1998) Expression of cyclin-dependent kinase 5 and associated cyclins in leydig and sertoli cells of the testis. J Androl 19:657–666
Negash S, Wang HS, Gao C, Ledee D, Zelenka P (2002) Cdk5 regulates cell–matrix and cell–cell adhesion in lens epithelial cells. J Cell Sci 115:2109–2117
Neystat M, Rzhetskaya M, Oo TF, Kholodilov N, Yarygina O, Wilson A, El-Khodor BF, Burke RE (2001) Expression of cyclin-dependent kinase 5 and its activator P35 in models of induced apoptotic death in neurons of the substantia nigra in vivo. J Neurochem 77(6):1611–1625
Nguyen MD, Lariviere RC, Julien JP (2001) Deregulation of Cdk5 in a mouse model of ALS: toxicity alleviated by perikaryal neurolament inclusions. Neuron 30:135–147
Nguyen KC, Rosales JL, Barboza M, Lee KY (2002a) Controversies over p25 in Alzheimer’s disease. J Alzheimers Dis 4:123–126
Nguyen MD, Mushynski WE, Julien JP (2002b) Cycling at the interface between neurodevelopment and neurodegeneration. Cell Death Differ 9:1294–1306
Nguyen MD, Boudreau M, Kriz J, Couillard-Despres S, Kaplan DR, Julien JP (2003) Cell cycle regulators in the neuronal death pathway of amyotrophic lateral sclerosis caused by mutant superoxide dismutase 1. J Neurosci 23:2131–2140
Nguyen MD, Julien JP (2003) Cyclin-Dependent kinase 5 in amyotrophic lateral sclerosis. Neurosignals 12:215–220
Nikolic M, Dudek H, Kwon YT, Ramos YFM, Tsai L-H (1996) The Cdk5/p35 kinase is essential for neurite outgrowth during neuronal differentiation. Genes Dev 7:816–825
Ohshima T, Ward JM, Huht C-G, Longenecker G, Veeranna, Pant HC, Brady RO, Martins LJ, Kulkarni AB (1996) Targeted disruption of the cyclin-dependent kinase 5 gene results in abnormal corticogenesis, neuronal pathology and perinatal death. Proc Natl Acad Sci 93(20):11173–11178
Paglini G, Peris L, Diez-Guerra J, Quiroga S, Cáceres A (2001a) The Cdk5-p35 kinase associates with the Golgi apparatus and regulates membrane traffic. EMBO Rep 2(12):1139–1144
Paglini G, Caceres A (2001b) The role of Cdk5-P35 kinase in neuronal development. Eur J Biochem 268(6):1528–1533
Pareek TK, Keller J, Kesavapany S, Pant HC, Iadarola MJ, Brady RO, Kulkarni AB (2006) Cdk5 activity regulates pain signaling. Proc Natl Acad Sci 103(3):791–796
Patrick GN, Zhou P, Kwon YT, Howley PM, Tsai L-H (1998) P35, the neuronal-specific activator of Cdk5 is degraded by the ubiquitin-proteasome pathway. J Biol Chem 273(37):24057–24064
Patrick GN, Zukerberg L, Nikolic M, Suzanne de la M, Dikkes P, Tsai L-H (1999) Conversion of P35 to P25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402:615–622
Patzke H, Maddineni U, Ayala R, Morabito M, Janet V, Dikkes P, Ahlijanian MK, Tsai L-H (2003) Partial rescue of the P35−/− brain phenotype by low expression of a neuronal-specific enolase P25 transgene. J Neurosci 23(7):2769
Paudel HK, Lew J, Ali Z, Wang JH (1993) Brain prolinedirected protein kinase phosphorylates tau on sites that are abnormally phosphorylated in tau associated with Alzheimer’s paired helical filaments. J Biol Chem 268:23512–23518
Philpott A, Porro EB, Kirschner MW, Tsai LH (1997) The role of Cdk5 and a novel regulatory subunit in regulating muscle differentiation and patterning. Genes Dev 11:1409–1421
Picconi B, Centonze D, Hakansson K, Bernardi G, Greengard P, Fisone G, Cenci MA, Calabresi P (2003) Loss of bidirectional striatal synaptic plasticity in l-DOPA-induced dyskinesia. Nat Neurosci 6:501–506
Rajgopal Y, Vemuri MC (2001) Ethanol induced changes in cyclin-dependent kinase-5 activity and its activators, P35, P67 (Munc-18) in rat brain. Neurosci Lett 308:173–176
Rakic S, Davis C, Molna ZN, Nikolic M, Parnavelas JG (2006) Role of p35/Cdk5 in preplate splitting in the developing cerebral cortex. Cereb Cortex 16:i35–i45
Rashid T, Banerjee M, Nikolic M (2001) Phosphorylation of Pak1 by the P35/Cdk5 kinase affects neuronal morphology. J Biol Chem 276(52):49043–49052
Rosales JL, Lee K-Y (2006) Extraneuronal roles of cyclin-dependent kinase 5. Bioessays 28(10):1023–1034
Rosales JL, Lee B-C, Modarressi M, Sarker KP, Lee K-Y, Jeong Y-G, Oko R, Lee K-Y (2004) Outer dense fibers (ODF) serve as a functional target for Cdk5.p35 in the developing sperm tail. J Biol Chem 279:1224–1232
Rosen DR, Siddique T, Patterson D et al (1993) Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362:59–62
Rimer M (2003) Neuregulins: primary or secondary signals for the control of synapse-specific gene expression. J Neurocytol 32:665–675
Sandal T, Stapnes C, Kleivdal H, Hedin L, Doskeland SO (2002) A novel, extraneuronal role for cyclin-dependent protein kinase 5 (CDK5): modulation of cAMP-induced apoptosis in rat leukemia cells. J Biol Chem 277:20783–20793
Session DR, Fautsch MP, Avula R, Jones WR, Nehra A et al (2001) Cyclin-dependent kinase 5 is expressed in both Sertoli cells and metaphase spermatocytes. Fertil Steril 75:669–673
Sharma P, Steinbach PJ, Sharma M, Amin ND, Barchi JJ Jr, Pant HC (1999a) Identification of substrate binding site of Cdk5. J Biol Chem 274(14):9600–9606
Sharma P, Sharma M, Amin ND, Albers WR, Pant HC (1999b) Regulation of cyclin-dependent kinase 5 catalytic activity by phosphorylation. Proc Natl Acad Sci 96(20):11156–11160
Sharma M, Sharma P, Pant HC (1999c) CDK-5-mediated neurofilament phosphorylation in SHSY5Y human neuroblastoma cells. J Neurochem 73:79–86
Shuang R, Zhang L, Fletcher A, Groblewski GE, Pevsner J, Stuenkel EL (1998) Regulation of Munc-18/syntaxin 1A interaction by cyclin-dependent kinase 5 in nerve endings. J Biol Chem 273(9):4957–4966
Shea TB, Yabe JT, Ortiz D, Pimenta A, Loomis P, Goldman RD, Amin N Pant HC (2003) Cdk5 regulates axonal transport and phosphorylation of neurofilaments in cultured neurons. J Cell Sci 117:933–941
Shelton SB, Johnson GVW (2004) Cyclin-dependent kinase-5 in neurodegeneration. J Neurochem 88(6):1313–1326
Shetty KT, Link WT, Pant HC (1993) cdc2-like kinase from rat spinal cord specifically phosphorylates KSPXK motifs in neurofilament proteins: isolation characterization. Proc Natl Acad Sci USA 90:6844
Shouqing L, Coralie V, Janet ED, David CR (2005) Cdk5 phosphorylation of Huntingtin reduces its cleavage by caspases: implications for mutant huntingtin toxicity. JCB 169(4):647–656
Smith DS, Tsai L-H (2002) Cdk5 the wheel: a role in trafficking and transport. Trends Cell Biol 12(1):28–36
Smith DS, Greer PL, Tsai L-H (2001) Cdk5 on the brain. Cell Growth Differ 12:277–283
Smith Deanna (2003) Cdk5 in neuroskeletal dynamics. Neurosignals 12:239–251
Smith PD, Crocker SJ, Jackson-Lewis V, Jordan-Sciutto KL, Hayley S, Mount MP, O’Hare MJ, Callaghan S, Slack RS, Przedborski S, Anisman H, Park DS (2003) Cyclin-dependent kinase 5 is a mediator of dopaminergic neuron loss in a mouse model of Parkinson’s disease. Proc Natl Acad Sci 100(23):13650–13655
Takahashi S, Saito T, Hisanaga S-i, Pant HC, Kulkarni Ashok B (2003) Tau phosphorylation by cyclin-dependent kinase 5/P39 during brain development reduces its affinity for microtubules. J Biol Chem 278(12):10506–10515
Tanaka T, Veeranna, Ohshima T, Rajan P, Amin ND, Cho A, Sreenath T, Pant HC, Brady RO, Kulkarni AB (2001) Neuronal Cdk5 activity is critical for survival. J Neurosci 21(2):550–558
Tang D, Yeung J, Lee KY, Matsushita M, Matsui H, Tomizawa K, Hatase O, Wang JH (1995) An isoform of the neuronal cyclin-dependent kinase 5 (Cdk5) activator. J Biol Chem 270:26897–26903
Timsit S, Menn B (2007) Cerebral ischemia, cell cycle elements and Cdk5. Biotechnol J 2(8):958–966
Tokouka H et al (2000) Brain-derived neurotrophic factorinduced phosphorylation of neurofilament-H subunit in primary cultures of embryo rat cortical neurons. J Cell Sci 113:1059–1068
Tomizawa K, Ohta J, Matsushita M, Moriwaki A, Li ST, Takei K, Matsui H (2002) Cdk5/P35 regulates neurotransmitter release through phosphorylation and downregulation of P/Q-type voltage-dependent calcium channel activity. J Neurosci 22:2590–2597
Tsai LH, Takahashi T, Caviness V, Harlow E (1993) Activity and expression pattern of cyclin-dependent kinase 5 in the embryonic mouse nervous system. Development 119:1029–1040
Tsai L-H (2003) The role of cyclin-dependent kinase 5 in brain development and degeneration. Cell Mol Biol Lett 8(2A):546–547
Uchida T, Ishiguro K, Ohnuma J, Takamatsu M, Yonekura S, Imahori K (1994) Precursor of cdk5 activator, the 23 kDa subunit of tau protein kinase II: its sequence and developmental change in brain. FEBS Lett 355:35–40
Van den Haute C, Spittaels K, Van Dorpe J, Lasrado R, Vandezande K, Laenen I, Geerts H, Van Leuven F (2000) Coexpression of human Cdk5 and its activator P35 with human protein tau in neurons in brain of triple transgenic mice. Neurobiol Dis 8(1):32–44
Vartiainen N, Keksa-Goldsteine V, Goldsteins G, Koistinaho J (2002) Aspirin provides cyclin-dependent kinase 5-dependent protection against subsequent hypoxia/reoxygenation damage in culture. J Neurochem 82:329–335
Walling AD (1999) Amyotrophic lateral sclerosis: Lou Gehrig’s disease. Am Fam Physician 59:1489–1496
Wang J, Liu S, Fu Y, Wang JH, Lu Y (2003) Cdk5 activation induces hippocampal CA1 cell death by directly phosphorylating NMDA receptors. Nat Neurosci 6(10):1039–1047
Wei FY, Nagashima K, Ohshima T, Saheki Y, Lu YF et al (2005) Cdk5-dependent regulation of glucose-stimulated insulin secretion. Nat Med 11:1104–1108
Weishaupt JH, Neusch C, Bähr M (2002) Cdk5 and neuronal cell death. Cell Tissue Res 312(1):1–8
Wong PC, Pardo CA, Borchelt DR, Lee MK, Copeland NG, Jenkins NA, Sisodia SS, Cleveland DW, Price DL (1995) An adverse property of a familial ALS-linked SOD1 mutation causes motor neuron disease characterized by vacuolar degeneration of mitochondria. Neuron 14:1105–1116
Xinrong F, Yuk-Kwan C, Dianbo Q, Yan Y, Nam SC, Robert ZQ (2006) Identification of nuclear import mechanisms for the neuronal Cdk5 activator. J Biol Chem 281(851):39014–39021
Yang HS, Hinds PW (2003) Increased Ezrin expression and activation by CDK5 coincident with acquisition of the senescent phenotype. Mol Cell 11:1163–1176
Yip YP, Capriotti C, Drill E, Tsai LH, Yip JW (2007) Cdk5 selectively affects the migration of different populations of neurons in the developing spinal cord. J Comp Neurol 503(2):297–307
Zhang Q, Ahuja HS, Zakeri ZF, Wolgemuth DJ (1997) Cyclin-dependent kinase 5 is associated with apoptotic cell death during development and tissue remodeling. Dev Biol 183:222–233
Zukerberg LR, Patrick GN, Nikolic M, Humbert S, Wu CL, Lanier LM, Gertler FB, Vidal M, Van Etten RA, Tsai LH (2000) Cables links Cdk5 and c-Abl and facilitates Cdk5 tyrosine phosphorylation, kinase upregulation, and neurite outgrowth. Neuron 26:633–646
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhariwala, F.A., Rajadhyaksha, M.S. An Unusual Member of the Cdk Family: Cdk5. Cell Mol Neurobiol 28, 351–369 (2008). https://doi.org/10.1007/s10571-007-9242-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-007-9242-1